Act like a helpful tutor and exlain me :6. Determine whether the following molecules are aromatic, antiaromatic, or nonaromatic. Justify your answers. (@ (b) © @ © [J @ ®)
Question:
Act like a helpful tutor and exlain me :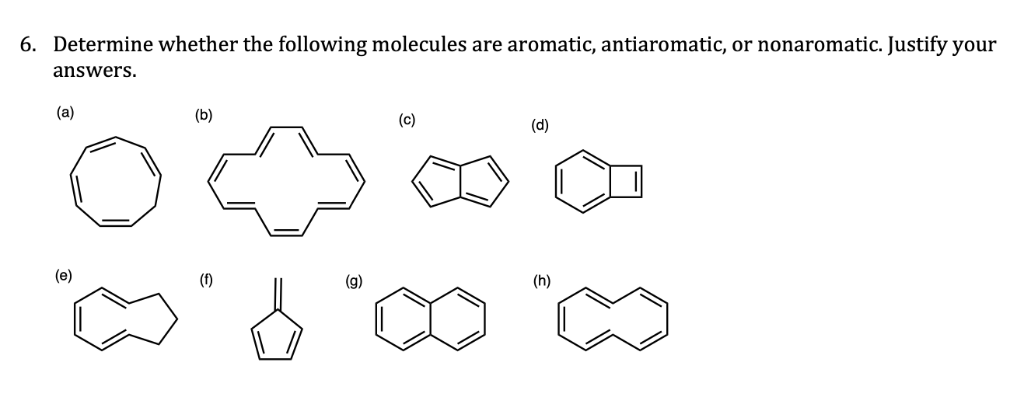 6. Determine whether the following molecules are aromatic, antiaromatic, or nonaromatic. Justify your
answers.
(@ (b) © @
© [J @ ®)
6. Determine whether the following molecules are aromatic, antiaromatic, or nonaromatic. Justify your
answers.
(@ (b) © @
© [J @ ®)
This is AI generated Response.
Asked by: Alok Kumar
Created at: 2026-02-03 05:01:28
Asked by: Alok Kumar
Created at: 2026-02-03 05:01:28
Final Answer:
Rendered
Raw Text
Did you like this answer?
# Aromaticity, Antiaromaticity, and Nonaromaticity
To determine if a molecule is **aromatic**, **antiaromatic**, or **nonaromatic**, use these criteria:
1. **Aromatic**:
- Cyclic
- Planar
- Fully conjugated (every atom in the ring has a p orbital)
- Follows **Hückel’s rule**: \(4n+2\) π electrons (where \(n = , 1, 2, \ldots\))
2. **Antiaromatic**:
- Cyclic
- Planar
- Fully conjugated
- **4n** π electrons
3. **Nonaromatic**:
- Not fully conjugated and/or not planar and/or not cyclic
---
## Analysis of Each Compound
### (a) Cyclooctatetraene (C₈H₈)
- **Structure:** 8-membered ring, 4 double bonds (8 π electrons)
- **Conjugation:** Although it has conjugated double bonds, it's *not planar* (adopts a tub-shaped conformation).
- **Conclusion:** **Nonaromatic**
- **Reason:** Not planar, not fully conjugated.
---
### (b) [10]-Annulene
- **Structure:** 10-membered ring, 5 double bonds (10 π electrons)
- **Conjugation:** In principle, it could be aromatic, but steric hindrance from internal hydrogens prevents planarity.
- **Conclusion:** **Nonaromatic**
- **Reason:** Not planar due to steric strain.
---
### (c) Indene
- **Structure:** Fused benzene and cyclopentene (6 + 2 = 8 π electrons, but cyclopentene is not aromatic)
- **Conjugation:** Benzene ring is aromatic, but the 5-membered ring is not fully conjugated.
- **Conclusion:** **Nonaromatic**
- **Reason:** The whole system is not fully conjugated.
---
### (d) Benzocyclobutadiene
- **Structure:** Benzene fused to cyclobutadiene
- **Conjugation:** Cyclobutadiene is antiaromatic (4 π electrons), benzene is aromatic (6 π electrons)
- **Conclusion:** **Nonaromatic**
- **Reason:** Fusion disrupts delocalization; system is not fully conjugated over both rings.
---
### (e) Azulene
- **Structure:** Fused 5- and 7-membered rings, 10 π electrons
- **Conjugation:** Fully conjugated and planar
- **Conclusion:** **Aromatic**
- **Reason:** Follows Hückel's rule (10 π electrons, n=2)
---
### (f) Cyclopentadienyl cation
- **Structure:** 5-membered ring, 2 double bonds, cation (4 π electrons)
- **Conjugation:** Planar, fully conjugated
- **Conclusion:** **Antiaromatic**
- **Reason:** 4n π electrons (n=1), follows antiaromatic rule
---
### (g) Naphthalene
- **Structure:** Two fused benzene rings, 10 π electrons
- **Conjugation:** Planar, fully conjugated
- **Conclusion:** **Aromatic**
- **Reason:** Follows Hückel's rule (10 π electrons, n=2)
---
### (h) Biphenylene
- **Structure:** Four fused benzene rings (12 π electrons)
- **Conjugation:** Planar, but 4n π electrons, destabilized
- **Conclusion:** **Antiaromatic**
- **Reason:** 4n π electrons (n=3); system is planar and fully conjugated
---
## **Summary Table**
| Molecule | Aromatic? | Reason |
|------------|------------------|---------------------------------------------|
| (a) | Nonaromatic | Not planar |
| (b) | Nonaromatic | Not planar |
| (c) | Nonaromatic | Not fully conjugated |
| (d) | Nonaromatic | Fusion disrupts conjugation |
| (e) | Aromatic | 10 π e⁻ (Hückel's rule), planar, conjugated |
| (f) | Antiaromatic | 4 π e⁻ (antiaromatic), planar, conjugated |
| (g) | Aromatic | 10 π e⁻ (Hückel's rule), planar, conjugated |
| (h) | Antiaromatic | 12 π e⁻ (antiaromatic), planar, conjugated |
---
**Let me know if you want to go deeper into any of these!**
📢 Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions