Act like a helpful tutor and exlain me :7. Draw the major and minor products for each reaction. Pay attention to stereochemistry. Indicate endo and exo products. If a reaction produces endo and exo racemic mixtures, show one enantiomer for each. ew + TU — [¢) + YN —_— F3C CFg, 0 De — [©] [e]
Question:
Act like a helpful tutor and exlain me :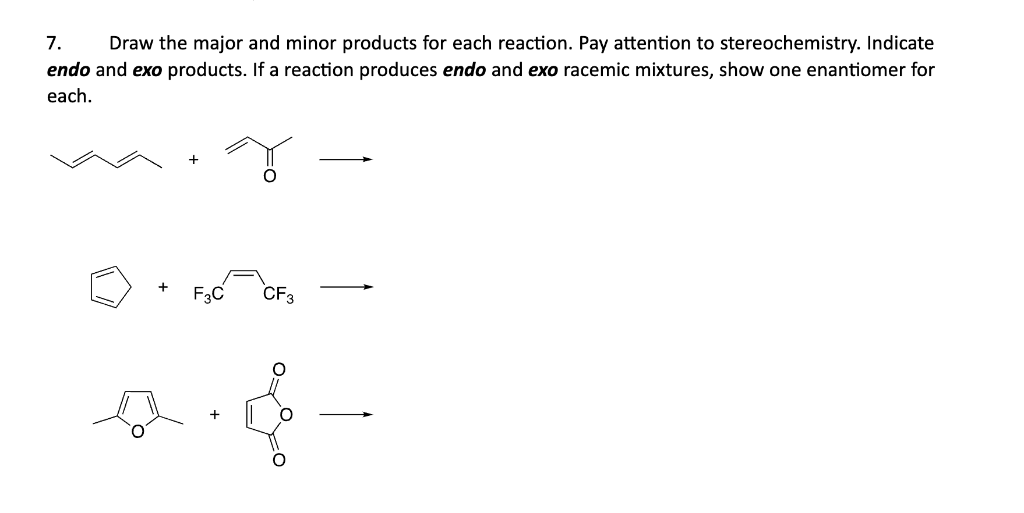 7. Draw the major and minor products for each reaction. Pay attention to stereochemistry. Indicate
endo and exo products. If a reaction produces endo and exo racemic mixtures, show one enantiomer for
each.
ew + TU —
[¢)
+ YN —_—
F3C CFg,
0
De —
[©]
[e]
7. Draw the major and minor products for each reaction. Pay attention to stereochemistry. Indicate
endo and exo products. If a reaction produces endo and exo racemic mixtures, show one enantiomer for
each.
ew + TU —
[¢)
+ YN —_—
F3C CFg,
0
De —
[©]
[e]
This is AI generated Response.
Asked by: Alok Kumar
Created at: 2026-01-29 06:56:15
Asked by: Alok Kumar
Created at: 2026-01-29 06:56:15
Final Answer:
Rendered
Raw Text
Did you like this answer?
Let's break down the question and each reaction:
---
## **What Type of Reaction?**
All three are **Diels-Alder reactions**.
A Diels-Alder reaction is a [4+2] cycloaddition between a diene (conjugated double bonds) and a dienophile (double or triple bond, often with electron-withdrawing groups).
- **Endo and Exo Products:**
- **Endo:** Substituents on the dienophile are oriented *under* the newly formed ring (towards the diene's π-system). Usually the major product due to secondary orbital interactions.
- **Exo:** Substituents are *away* from the π-system (above the plane).
---
## **1. 1,3-Butadiene + Methyl Vinyl Ketone**
**Structures:**
- Diene: 1,3-Butadiene
- Dienophile: Methyl vinyl ketone (CH₂=CH-COCH₃)
**Product:**
A cyclohexene ring forms, with a ketone at the 3-position and a methyl at the 4-position (numbering starts at the top of the diene).
### **Endo Product (Major)**
- The carbonyl group is oriented under the ring.
- Draw one enantiomer if chiral.
### **Exo Product (Minor)**
- The carbonyl group is oriented away from the ring.
---
## **2. Benzene + Hexafluoro-2-butyne**
**Note:** Benzene does **not** do Diels-Alder reactions; it is too stable (aromatic).
But if you meant **furan** or **cyclopentadiene**, those would react. If this is benzene, **no reaction** occurs.
---
## **3. 2,5-Dimethylfuran + Maleic Anhydride**
- Diene: 2,5-Dimethylfuran
- Dienophile: Maleic anhydride
### **Endo Product (Major)**
- The anhydride (two carbonyls) are oriented under the ring.
- Show one enantiomer if chiral.
### **Exo Product (Minor)**
- The anhydride is oriented away from the ring.
---
## **Summary Table**
| Reaction | Major Product | Minor Product | Endo/Exo | Stereochemistry? |
|----------|--------------|--------------|----------|------------------|
| 1 | Endo | Exo | Yes | Show one enantiomer for each if chiral |
| 2 | No reaction | - | - | - |
| 3 | Endo | Exo | Yes | Show one enantiomer for each if chiral |
---
## **Drawing the Products**
### **How to Draw:**
- Draw the cyclohexene ring for Diels-Alder.
- Attach substituents at correct positions.
- For endo: place EWG (like C=O, anhydride) *under* the ring.
- For exo: place EWG *above* the ring.
- If chiral, show wedge/dash for stereochemistry.
---
## **Example for 1st Reaction:**
- **Endo (major):**
- Cyclohexene ring
- COCH₃ group under the ring (endo)
- Methyl group
- **Exo (minor):**
- COCH₃ group above the ring (exo)
- Methyl group
---
If you want to see the actual structures drawn out, let me know! I can help sketch them or explain the step-by-step drawing.
📢 Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions