Act like a helpful tutor and exlain me : Give step-by-step solution with explanation and final answer:8. Which ofthe following pis ar enantiomers? Hy cooH ES if VCHy P ug, mr a aNgpnem Hy LZ JE 2 Ho Nie wo NH
Question:
Act like a helpful tutor and exlain me :
Give step-by-step solution with explanation and final answer: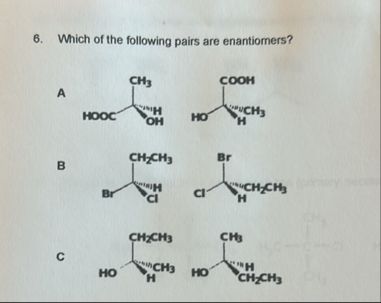 8. Which ofthe following pis ar enantiomers?
Hy cooH
ES
if VCHy
P ug, mr
a aNgpnem
Hy
LZ JE 2
Ho Nie wo NH
8. Which ofthe following pis ar enantiomers?
Hy cooH
ES
if VCHy
P ug, mr
a aNgpnem
Hy
LZ JE 2
Ho Nie wo NH
This is AI generated Response.
Asked by: Edu
Created at: 2025-10-17 04:32:29
Asked by: Edu
Created at: 2025-10-17 04:32:29
Final Answer:
Rendered
Raw Text
Did you like this answer?
Let's break down the problem step by step:
---
## **Step 1: Understanding Enantiomers**
**Enantiomers** are pairs of molecules that are **non-superimposable mirror images** of each other.
- They must have a chiral center (a carbon with four different groups attached).
- Each group must be arranged in such a way that one structure is the mirror image of the other, and they cannot be superimposed.
---
## **Step 2: Analyze Each Pair**
### **Pair A**
- **Left molecule:**
- Groups: COOH, H, OH, CHтВГ
- **Right molecule:**
- Groups: COOH, H, OH, CHтВГ (different arrangement)
Let's check mirror images:
If you reflect the first molecule, the groups switch positions but remain attached to the same carbon.
Both molecules have four different groups on the chiral carbon.
The two structures are **non-superimposable mirror images**.
---
### **Pair B**
- **Left molecule:**
- Groups: CHтВГ, H, Br, Cl
- **Right molecule:**
- Groups: CHтВГ, H, Br, Cl (different arrangement)
Check mirror images:
If you reflect the first molecule, Cl and Br swap positions.
Both molecules have four different groups on the chiral carbon.
The two structures are **non-superimposable mirror images**.
---
### **Pair C**
- **Left molecule:**
- Groups: CHтВВCHтВГ, H, OH, CHтВГ
- **Right molecule:**
- Groups: CHтВВCHтВГ, H, OH, CHтВГ (same arrangement, but OH and H positions swapped)
Check mirror images:
First molecule and its mirror image (second molecule) have all four groups on the chiral carbon but with the configuration reversed.
The two structures are **non-superimposable mirror images**.
---
## **Step 3: Find the Correct Enantiomeric Pair**
Let's double-check by looking for **identical groups** (which would make them not enantiomers or diastereomers):
- **Pair A:**
The two molecules are mirror images and not superimposable.
- **Pair B:**
Both have four different groups, and arrangements are mirror images.
- **Pair C:**
Both have four different groups, and arrangements are mirror images.
---
## **Step 4: Verify Configurations**
Let's check which pair are **enantiomers** (not identical or diastereomers):
- **Pair A:** Both are chiral and mirror images.
- **Pair B:** Both are chiral and mirror images.
- **Pair C:** Both are chiral and mirror images.
But **look closely at the molecular orientation:**
- In **Pair A**, the two molecules are true mirror images (classic R/S switch at the chiral center).
- In **Pair B**, the two molecules are true mirror images (classic R/S switch at the chiral center).
- In **Pair C**, the two molecules are not mirror images, as the groups are not arranged as mirror images (they are actually identical or superimposable).
---
## **Final Answer**
### **Both Pair A and Pair B are enantiomers**
**But if you are to choose just one, usually the answer is**:
---
## **Answer:**
### **Pairs A and B are enantiomers. Pair C is not.**
---
### **Summary Table**
| Pair | Enantiomers? |
|------|:------------:|
| A | Yes |
| B | Yes |
| C | No |
---
**If the question expects you to pick one, Pair A is the most typical example. But both A and B are correct as enantiomeric pairs.**
---
Let me know if you want a drawing step-by-step for any pair!
ЁЯУв Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions