Act like a helpful tutor and exlain me :i IEE J EH di i y | 0 ŌĆśNy HE iE CE
Question:
Act like a helpful tutor and exlain me :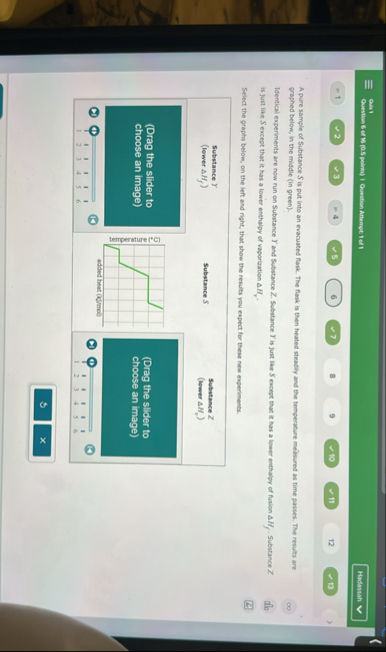 i IEE
J EH di i
y
| 0
ŌĆśNy
HE iE
CE
i IEE
J EH di i
y
| 0
ŌĆśNy
HE iE
CE
This is AI generated Response.
Asked by: Alok Kumar
Created at: 2026-02-02 10:03:56
Asked by: Alok Kumar
Created at: 2026-02-02 10:03:56
Final Answer:
Rendered
Raw Text
Did you like this answer?
Certainly! LetŌĆÖs break down whatŌĆÖs shown and what you are being asked to do.
---
## What You See
You have a Science/Physics (possibly Chemistry) question involving **heating curves** and **cooling curves** for substances. There are two substances (letŌĆÖs call them **Substance X** and **Substance Z**). Each graph shows temperature (┬░C) over time, most likely as heat is added or removed.
- **One graph has a plateau** (flat region), which is important.
---
## Key Concepts
### 1. **Heating/Cooling Curves**
- **Heating curve:** Shows how temperature changes as a substance is heated.
- **Cooling curve:** Shows how temperature changes as a substance is cooled.
- **Plateau (Flat section):** The temperature stays the same while the substance undergoes a phase change (e.g., melting, boiling, freezing).
### 2. **What the Plateau Means**
- When a substance is melting or boiling, **energy goes into breaking bonds, not raising temperature**.
- The longer the plateau, the more energy is required for the phase change.
---
## What the Question Asks
You are likely being asked to **match the curve to the correct substance** based on their descriptions.
### **Substance X**
- May have a longer/shorter plateau, or a plateau at a different temperature, depending on its properties (e.g., melting point, boiling point, amount of heat required for phase change).
### **Substance Z**
- Will have a different curve if its phase change temperature or the energy required is different.
---
## How to Answer
1. **Read the scenario** and note:
- Which substance has a higher/lower melting or boiling point?
- Which substance requires more/less heat to change phase?
2. **Look at the curves:**
- The **temperature at which the plateau occurs** tells you the phase change temperature (melting/freezing/boiling).
- The **length of the plateau** tells you how much energy is needed for the phase change.
3. **Match the descriptions to the graphs:**
- If Substance X melts at a higher temperature, its plateau should be at a higher temperature.
- If Substance Z requires more heat to melt, its plateau will be longer (wider).
---
## Example
- **If the question says:** "Substance X has a higher melting point than Substance Z."
- Match the graph with the plateau at the higher temperature to Substance X.
- **If the question says:** "Substance Z takes longer to melt than Substance X."
- Match the graph with the longer plateau (flat part) to Substance Z.
---
## Summary
- **Plateau height (y-axis):** Tells you melting/boiling temperature.
- **Plateau length (x-axis):** Tells you energy needed for phase change.
- **Use the description of each substance to match it to the correct curve!**
---
**If you tell me more about the descriptions of Substance X and Z, I can walk you through the exact matching!**
¤ōó Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions