Answers07. Alkenes that are more substituted (i.e., have more carbons attached to the C=C double bond) are more electron rich and react faster with electrophilic reagents (e.g., HCI). The addition of HCI to cis-2- butene (1) or trans-2-butene (2), both of which have the same substitution, gives the same product, 2- chlorobutane (3). In the scheme below, circle the 2-butene isomer that reacts faster in this reaction. Hint: Draw the potential energy (PE) vs reaction coordinate (RC) diagram you saw for the Sy1 reaction (or review the University of Surrey video we saw in class). Are the isomeric 2-butenes at the same energy level in the starting material? Which one is higher? (06 points) HG CHy HCI W Hol HQ A REEL SE EP TE i BB © Ky HC ko CH 1 3 2 08. The reagent phenylsulfenyl chloride (PhSC) is known to add to alkenes in a stereospecific anti fashion (like the halogens). Given that the exo (top) face of bridged bicyclic alkenes is less hindered than the corresponding endo (bottom) face, predict the product of the following reaction. Hint: S is less electronegative than CI. Build a molecular model to help! (06 points) A PhSCI 09. Configuration. For each stereogenic center in the following chiral molecules, give the proper configuration(s) using the R/S (i.e., Cahn-Ingold-Prelog) convention. Please write only "R" or "S" in the space to the right of each structure. Hint: Use molecular models to help your with your responses! (08 points - 02 points each) a. b. woh \ fiber CoH HNC SC oli pl ofan as 9 N=7 | 3 y Cel 8 i A 0=9 3 de PSA R=\ HCS UD sH c H ie Ts A S= 1b G=0 H=\ C= p 11
Question:
Answers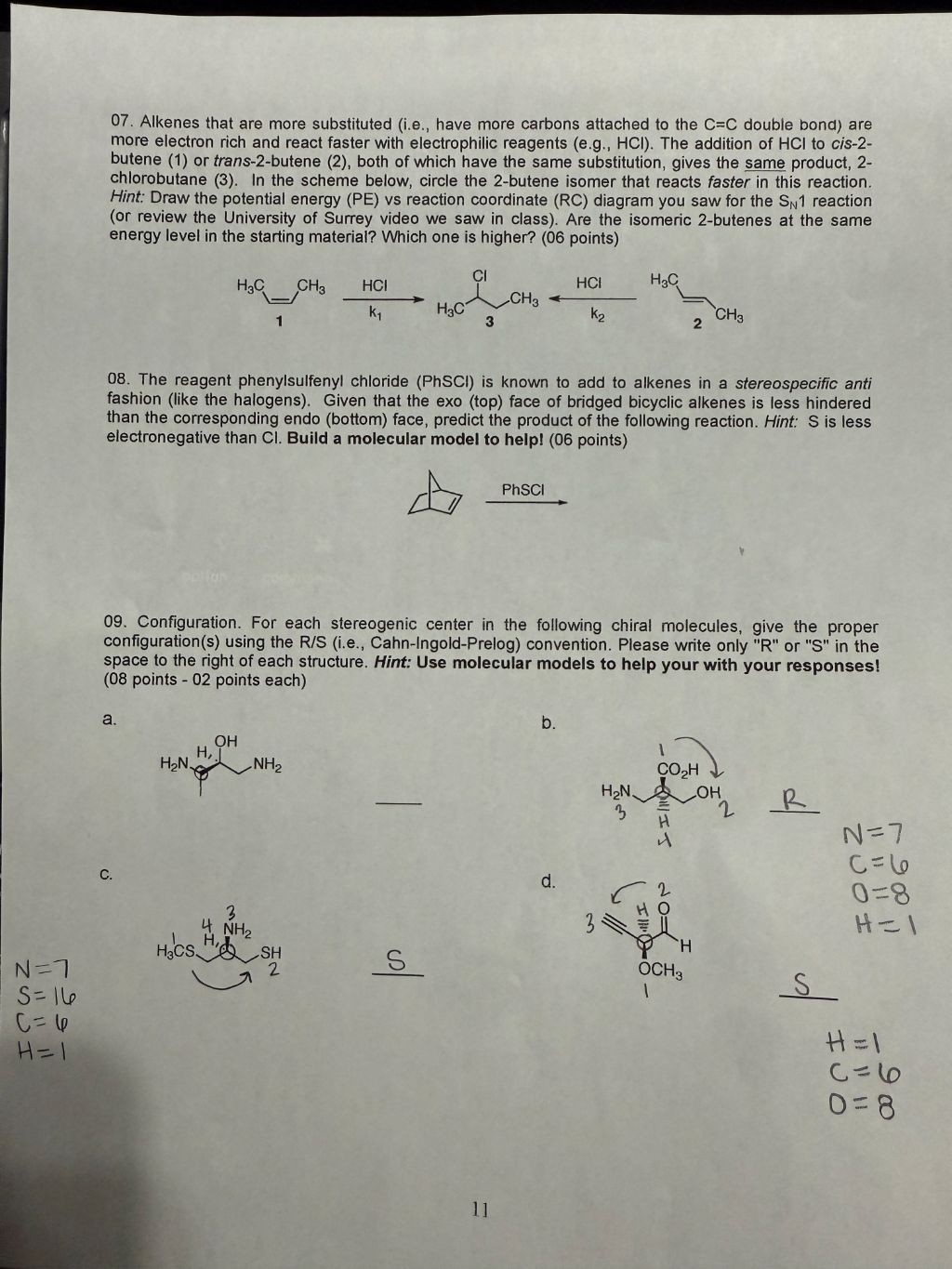 07. Alkenes that are more substituted (i.e., have more carbons attached to the C=C double bond) are
more electron rich and react faster with electrophilic reagents (e.g., HCI). The addition of HCI to cis-2-
butene (1) or trans-2-butene (2), both of which have the same substitution, gives the same product, 2-
chlorobutane (3). In the scheme below, circle the 2-butene isomer that reacts faster in this reaction.
Hint: Draw the potential energy (PE) vs reaction coordinate (RC) diagram you saw for the Sy1 reaction
(or review the University of Surrey video we saw in class). Are the isomeric 2-butenes at the same
energy level in the starting material? Which one is higher? (06 points)
HG CHy HCI W Hol HQ
A REEL SE EP TE i BB ©
Ky HC ko CH
1 3 2
08. The reagent phenylsulfenyl chloride (PhSC) is known to add to alkenes in a stereospecific anti
fashion (like the halogens). Given that the exo (top) face of bridged bicyclic alkenes is less hindered
than the corresponding endo (bottom) face, predict the product of the following reaction. Hint: S is less
electronegative than CI. Build a molecular model to help! (06 points)
A PhSCI
09. Configuration. For each stereogenic center in the following chiral molecules, give the proper
configuration(s) using the R/S (i.e., Cahn-Ingold-Prelog) convention. Please write only "R" or "S" in the
space to the right of each structure. Hint: Use molecular models to help your with your responses!
(08 points - 02 points each)
a. b.
woh \
fiber CoH
HNC SC oli pl
ofan as
9 N=7
| 3 y Cel
8 i A 0=9
3 de PSA R=\
HCS UD sH c H
ie Ts A
S= 1b
G=0
H=\
C=
p 11
07. Alkenes that are more substituted (i.e., have more carbons attached to the C=C double bond) are
more electron rich and react faster with electrophilic reagents (e.g., HCI). The addition of HCI to cis-2-
butene (1) or trans-2-butene (2), both of which have the same substitution, gives the same product, 2-
chlorobutane (3). In the scheme below, circle the 2-butene isomer that reacts faster in this reaction.
Hint: Draw the potential energy (PE) vs reaction coordinate (RC) diagram you saw for the Sy1 reaction
(or review the University of Surrey video we saw in class). Are the isomeric 2-butenes at the same
energy level in the starting material? Which one is higher? (06 points)
HG CHy HCI W Hol HQ
A REEL SE EP TE i BB ©
Ky HC ko CH
1 3 2
08. The reagent phenylsulfenyl chloride (PhSC) is known to add to alkenes in a stereospecific anti
fashion (like the halogens). Given that the exo (top) face of bridged bicyclic alkenes is less hindered
than the corresponding endo (bottom) face, predict the product of the following reaction. Hint: S is less
electronegative than CI. Build a molecular model to help! (06 points)
A PhSCI
09. Configuration. For each stereogenic center in the following chiral molecules, give the proper
configuration(s) using the R/S (i.e., Cahn-Ingold-Prelog) convention. Please write only "R" or "S" in the
space to the right of each structure. Hint: Use molecular models to help your with your responses!
(08 points - 02 points each)
a. b.
woh \
fiber CoH
HNC SC oli pl
ofan as
9 N=7
| 3 y Cel
8 i A 0=9
3 de PSA R=\
HCS UD sH c H
ie Ts A
S= 1b
G=0
H=\
C=
p 11
Asked by: Chirag
Created at: 2025-07-29 04:17:28
Final Answer:
📢 Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions