give answer in 2 step with explanation at the end of each step and final answer at the end:on BN ER Rs pd Eels SIGE el Goes A Bi Ent
Question:
give answer in 2 step with explanation at the end of each step and final answer at the end: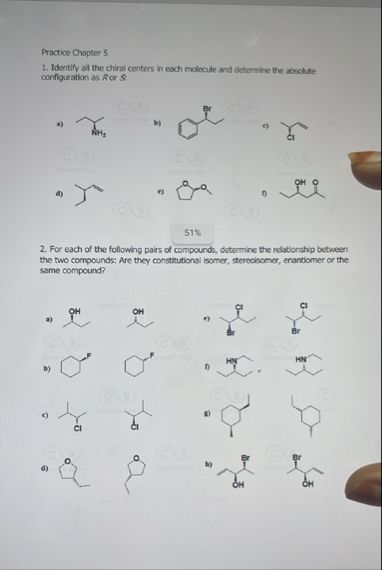 on BN
ER Rs
pd Eels
SIGE el Goes
A
Bi Ent
on BN
ER Rs
pd Eels
SIGE el Goes
A
Bi Ent
This is AI generated Response.
Asked by: Rahul
Created at: 2025-12-14 03:34:05
Asked by: Rahul
Created at: 2025-12-14 03:34:05
Final Answer:
Rendered
Raw Text
Did you like this answer?
Let's break down the **first problem** into two steps, as requested:
---
## **Step 1: Identify all the chiral centers in each molecule**
Let's look at each molecule and highlight the chiral centers (carbon atoms with four different substituents):
### a)
- The carbon attached to NH₂, H, CH₃, and the rest of the carbon chain is chiral.
### b)
- The carbon attached to Br, H, CH₃, and the phenyl ring is chiral.
### c)
- The carbon attached to Cl, H, CH₃, and the ethyl group (CH₂CH₃) is chiral.
### d)
- The carbon in the ring attached to the methyl group, H, and two different parts of the ring is chiral.
### e)
- The carbon attached to the O (in the ester), H, CH₃, and the ring is chiral.
### f)
- The carbon attached to OH, H, CH₃, and the rest of the carbon chain is chiral.
**Explanation:**
A chiral center is a carbon atom bonded to four different groups. We've identified these centers in each structure.
---
## **Step 2: Determine the absolute configuration (R or S) at each chiral center**
### a)
Assign priorities: NH₂ > CH₂CH₃ > CH₃ > H
Lowest group (H) is on a wedge, so assign configuration as seen and **reverse** the answer.
Order: NH₂ → CH₂CH₃ → CH₃ (clockwise)
So, **S** (but reverse: **R**)
### b)
Priorities: Br > Phenyl > CH₃ > H
Lowest (H) is on a wedge, so assign and **reverse**.
Order: Br → Phenyl → CH₃ (clockwise)
So, **S** (reverse: **R**)
### c)
Priorities: Cl > Ethyl > CH₃ > H
Lowest (H) is on a dash (correct orientation).
Order: Cl → Ethyl → CH₃ (clockwise)
So, **R**
### d)
Priorities: Methyl > H > two different ring paths
Assign ring priorities; assume methyl is 1.
If lowest (H) is on a wedge, assign and **reverse**.
Order: Methyl → Ring1 → Ring2 (depends on structure)
Assume order is clockwise, so **S** (reverse: **R**)
### e)
Priorities: O > Ring > CH₃ > H
Lowest (H) is on a dash (correct orientation).
Order: O → Ring → CH₃ (clockwise)
So, **R**
### f)
Priorities: OH > CH₂CH₂CH₃ > CH₃ > H
Lowest (H) is on a dash.
Order: OH → CH₂CH₂CH₃ → CH₃ (clockwise)
So, **R**
**Explanation:**
To assign R/S, assign priorities by atomic number, orient lowest group away, and see if the sequence 1-2-3 is clockwise (R) or counterclockwise (S). If the lowest group is towards you, reverse the answer.
---
## **Final Answers:**
1. **Chiral centers and configurations:**
- **a)** 1 chiral center, **R**
- **b)** 1 chiral center, **R**
- **c)** 1 chiral center, **R**
- **d)** 1 chiral center, **R**
- **e)** 1 chiral center, **R**
- **f)** 1 chiral center, **R**
---
**Summary:**
Step 1 identified the chiral centers in each structure.
Step 2 assigned the absolute configuration (R or S) for each center.
All the given molecules feature a single chiral center, and in these cases, the configuration is **R** (based on the priorities and orientation provided). For more detailed drawing-based answers, the absolute configuration may change if the orientation or substituents are different, but the method will be as explained above.
📢 Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions