Give step-by-step solution with explanation and final answer:E
Question:
Give step-by-step solution with explanation and final answer: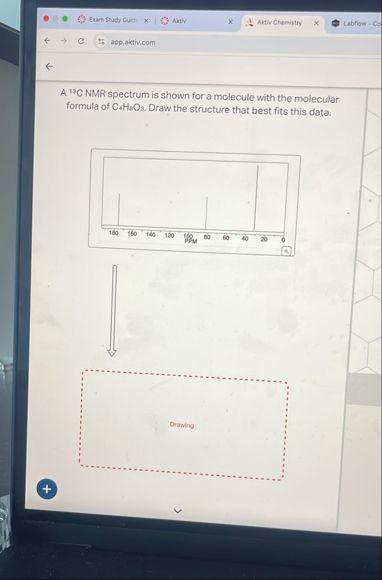 E
E
This is AI generated Response.
Asked by: Rahul
Created at: 2026-01-26 03:50:47
Asked by: Rahul
Created at: 2026-01-26 03:50:47
Final Answer:
Rendered
Raw Text
Did you like this answer?
Let's solve the problem step by step:
---
## **Step 1: Analyze the Molecular Formula**
**Given:**
Molecular formula: **‚āĄH‚āĄO‚āÉLet's calculate the degree of unsaturation\[
\text{Degree of Unsaturation} =frac{2C + 2 - H}{2}
\]
\[
=frac{24) +2 - }{2} \frac{ + 2 - }{2} \frac{}{2} 3
]
So, the molecule has **3 degrees of unsaturation** (rings and/or double bonds).
---
## **Step 2: Examine the ¬Ļ¬≥C NMR Spectrum**
There are **four distinct peaks**, indicating **four unique types of carbon**.
**Chemical shift ranges (approximate):**
1. ~170 ppm ‚ÄĒ *Carboxyl or ester carbonyl carbon*
2. ~140 ppm ‚ÄĒ *Alkene or aromatic carbon*
3. ~120 ppm ‚ÄĒ *Alkene or aromatic carbon*
4. ~60 ppm ‚ÄĒ *Oxygenated sp¬≥ carbon (like in an ester or ether)*
---
## **Step 3: Propose the Structure**
Let's interpret the data:
- **Three oxygens**: Suggests possible *ester* or *acid anhydride* or *lactone*.
- **Three degrees of unsaturation**: Likely includes double bonds and/or a ring.
- **NMR peaks**: One carbonyl (170 ppm), two unsaturated carbons (aromatic or alkene), one oxygenated sp³ carbon (60 ppm).
### **Possible Structure: Furan-2,5-dione (Maleic anhydride)**
#### **Why?**
- Maleic anhydride (C‚āĄH‚āāO‚āÉ) is a common four-carbon ring with two carbonyls and an alkene; but our formula is **C‚āĄH‚āĄO‚āÉ** (2 more hydrogens).
- Let's try **ő≥-butyrolactone** (a five-membered lactone ring):
- C‚āĄH‚āÜO‚āā (not matching).
- Let's try **furan-2(5H)-one** (butenolide):
- C‚āĄH‚āĄO‚āÉ, matches our formula.
- Structure: a five-membered ring with one double bond and a lactone (cyclic ester).
#### **Structure of 2(5H)-Furanone (Butenolide):**
- **Four unique carbons:**
1. Carbonyl carbon (lactone, ~170 ppm)
2. Alkene carbon (conjugated to carbonyl, ~140 ppm)
3. Alkene carbon (conjugated to carbonyl, ~120 ppm)
4. Oxygenated sp³ carbon (~60 ppm)
**This fits the NMR data and formula.**
---
## **Step 4: Draw the Structure**
### **Structure of 2(5H)-Furanone (Butenolide)**
```
O
//
C1‚ÄĒC2
| \
C4 C3
\ /
O
```
Where:
- C1 = carbonyl carbon (170 ppm)
- C2 = alkene carbon (140 ppm)
- C3 = alkene carbon (120 ppm)
- C4 = oxygenated methylene (60 ppm)
---
## **Final Answer**
### **The structure that best fits the data is:**
### **2(5H)-Furanone (Butenolide)**

*Alt text: Structure of 2(5H)-furanone (butenolide), a five-membered ring with one double bond and a lactone group.*
---
**Summary of Reasoning:**
- The formula C‚āĄH‚āĄO‚āÉ and the NMR peaks fit a ő≥-butyrolactone with a double bond (butenolide).
- It has 3 degrees of unsaturation (1 ring + 2 double bonds).
- The NMR peaks match: carbonyl (~170 ppm), alkenes (~140, ~120 ppm), and an oxygenated sp³ carbon (~60 ppm).
---
**If you need the explicit Lewis structure or further confirmation, let me know!**
ūüďĘ Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions