Give step-by-step solution with explanation and final answer:| SnBuy Nn Pd(PPh;), Ssi AL io toluene, 80 °C Br 0 1. Bu,SnCl —_— | N . { { Br F Pd(PPh;), Draw the molecule on the canvas by choos Br Bu;Sn Pd(PPh,), N toluene
Question:
Give step-by-step solution with explanation and final answer: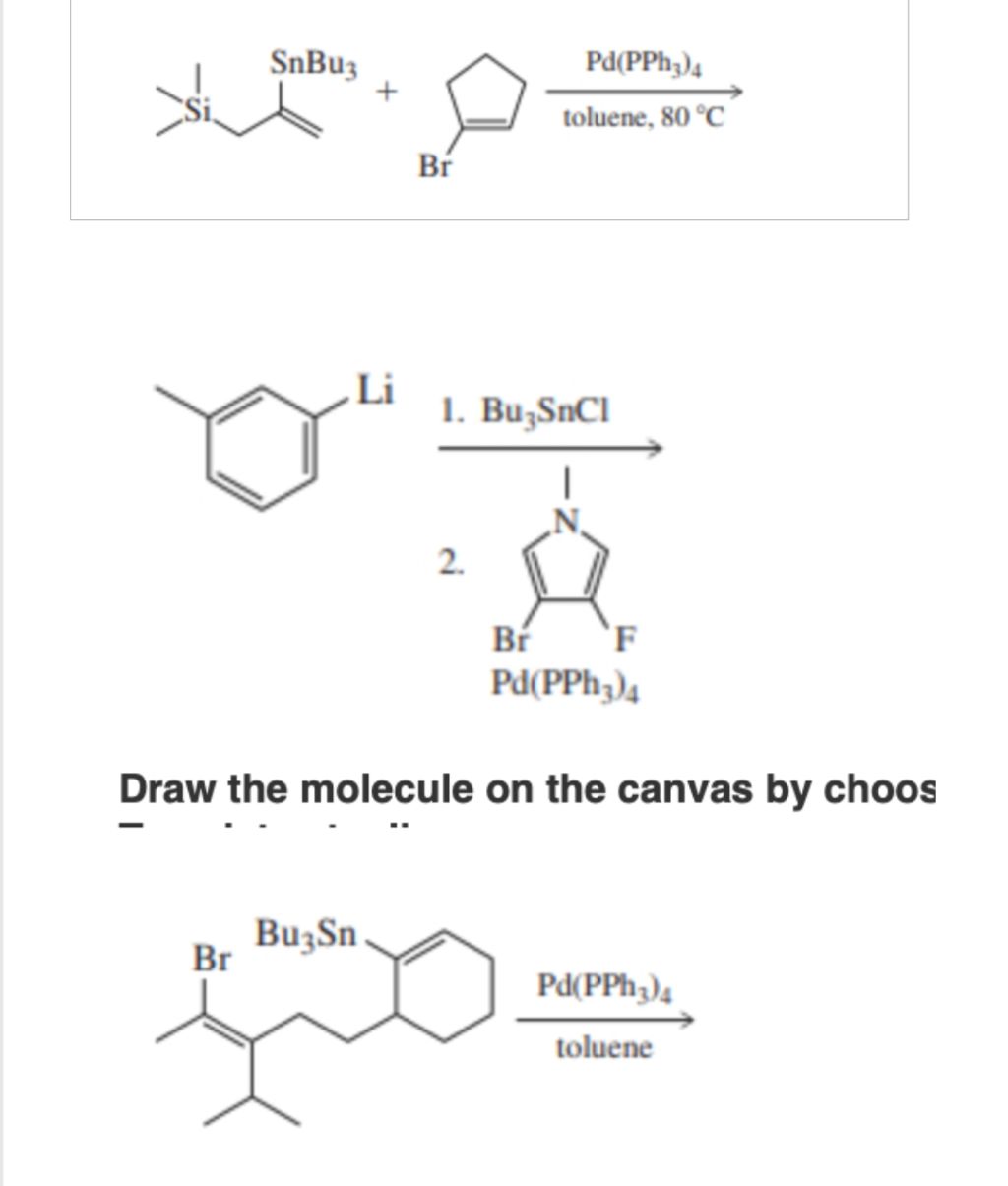 | SnBuy Nn Pd(PPh;),
Ssi AL io toluene, 80 °C
Br
0 1. Bu,SnCl
—_—
|
N
. { {
Br F
Pd(PPh;),
Draw the molecule on the canvas by choos
Br Bu;Sn
Pd(PPh,),
N toluene
| SnBuy Nn Pd(PPh;),
Ssi AL io toluene, 80 °C
Br
0 1. Bu,SnCl
—_—
|
N
. { {
Br F
Pd(PPh;),
Draw the molecule on the canvas by choos
Br Bu;Sn
Pd(PPh,),
N toluene
This is AI generated Response.
Asked by: Om
Created at: 2026-02-01 06:49:21
Asked by: Om
Created at: 2026-02-01 06:49:21
Final Answer:
Rendered
Raw Text
Did you like this answer?
Let's solve each reaction step by step. All of these reactions involve the **Stille coupling**, a palladium-catalyzed cross-coupling between an organostannane (R-SnBu₃) and an organohalide (R'-X).
---
## **1. First Reaction**
### **Given:**
- **Reactants:**
- (TMS-alkene)-SnBu₃
- Bromocyclopentadiene
- **Catalyst:** Pd(PPh₃)₄, toluene, 80°C
### **Step-by-Step Solution:**
1. **Identify Stille Coupling:**
The organostannane (R-SnBu₃) reacts with the aryl/vinyl halide (R'-Br) in the presence of Pd() catalyst.
2. **Coupling:**
The SnBu₃ group is replaced by the aryl/vinyl group from the halide, forming a new C–C bond.
### **Product:**
- The double bond (alkene) with the TMS group is now attached to the cyclopentadiene ring (where Br was).
- Final product:
```
(TMS-alkene)-cyclopentadiene
```
---
## **2. Second Reaction**
### **Given:**
- **Step 1:** p-Tolyl lithium + Bu₃SnCl → p-Tolyl-SnBu₃
- **Step 2:** p-Tolyl-SnBu₃ + 2-bromo-4-fluoropyrrole (Pd catalyst)
### **Step-by-Step Solution:**
1. **Transmetallation:**
The p-tolyl lithium reacts with Bu₃SnCl to give p-tolyl-SnBu₃.
2. **Stille Coupling:**
This organostannane reacts with bromo-fluoropyrrole, attaching the p-tolyl group to the pyrrole where Br was.
### **Product:**
- p-Tolyl attached to pyrrole at the 2-position (where Br was), with F still at the 4-position.
---
## **3. Third Reaction**
### **Given:**
- **Reactant:** A molecule with both Br and SnBu₃ groups, plus a fused ring system (decalin derivative).
- **Catalyst:** Pd(PPh₃)₄, toluene
### **Step-by-Step Solution:**
1. **Intramolecular Stille Coupling:**
The Br and SnBu₃ are on the same molecule. Under Pd() catalysis, they form a new C–C bond, closing a ring.
### **Product:**
- A new fused ring is formed, connecting the carbons originally bearing Br and SnBu₃.
---
## **Summary Table**
| Step | Type of Reaction | Main Transformation | Product Sketch |
|------|---------------------|------------------------------------------|---------------------------------------------------------------------|
| 1 | Stille Coupling | Attach TMS-alkene to cyclopentadiene |  |
| 2 | Stille Coupling | Attach p-tolyl to 2-position of pyrrole |  |
| 3 | Intramolecular Stille | Form new ring (close the molecule) |  |
---
## **Final Answers (in words):**
### 1. **(TMS-alkene)-cyclopentadiene**
The alkene chain with a TMS group is attached to cyclopentadiene at the position where Br was.
### 2. **p-Tolyl-2-fluoropyrrole**
The p-tolyl group is attached to the 2-position of the pyrrole ring (where Br was), F remains at the 4-position.
### 3. **Fused bicyclic ring**
The molecule forms a new C–C bond between the formerly bromo and stannyl groups, resulting in a fused bicyclic system.
---
**If you need the explicit structures drawn out, let me know!**
📢 Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions