Give step-by-step solution with explanation and final answer:Em ey aA SLEESES Or pr SI [EŌĆö, a ŌĆ£ co EE. Co Cra Eo ŌĆ£To J el BR cee A
Question:
Give step-by-step solution with explanation and final answer: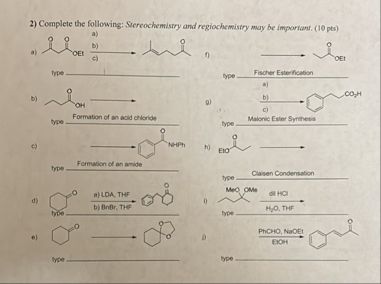 Em ey
aA SLEESES Or pr
SI [EŌĆö,
a ŌĆ£ co
EE. Co Cra
Eo
ŌĆ£To J
el
BR cee A
Em ey
aA SLEESES Or pr
SI [EŌĆö,
a ŌĆ£ co
EE. Co Cra
Eo
ŌĆ£To J
el
BR cee A
This is AI generated Response.
Asked by: Rahul
Created at: 2026-02-12 16:46:24
Asked by: Rahul
Created at: 2026-02-12 16:46:24
Final Answer:
Rendered
Raw Text
Did you like this answer?
Let's solve each reaction step-by-step, identifying the reaction type and providing the final product.
---
## a)
**Given:**
- Reactant:  (ethyl acetoacetate)
- Reagent: \( \mathrm{Br_2, NaOEt} \)
- Product: ?
**Type:** ╬▒-Bromination
### **Step-by-step:**
1. **NaOEt** deprotonates the ╬▒-hydrogen (adjacent to the carbonyl), forming an enolate.
2. **BrŌéé** reacts with the enolate, brominating the ╬▒-position.
**Final answer:**
- Product: 
- Structure:

- Type: ╬▒-Bromination
---
## b)
**Given:**
- Reactant: Carboxylic acid
- Reagent: SOClŌéé
- Product: ?
**Type:** Formation of an acid chloride
### **Step-by-step:**
1. **SOClŌéé** converts carboxylic acid to acid chloride.
**Final answer:**
- Product: Acid chloride (replace OH with Cl)
- Type: Formation of an acid chloride
---
## c)
**Given:**
- Reactant: Acid chloride
- Reagent: Amine (with aromatic ring)
- Product: ?
**Type:** Formation of an amide
### **Step-by-step:**
1. **Aromatic amine** attacks the acid chloride, forming an amide.
**Final answer:**
- Product: Amide (replace Cl with NHR, where R is the aromatic group)
- Type: Formation of an amide
---
## d)
**Given:**
- Reactant: Cyclohexanone
- Reagents:
1. LDA, THF
2. Benzyl bromide
**Type:** Alkylation of Enolate
### **Step-by-step:**
1. **LDA** forms an enolate at the ╬▒-position.
2. The enolate attacks benzyl bromide, alkylating the ╬▒-carbon.
**Final answer:**
- Product: Cyclohexanone with a benzyl group at the ╬▒-position.
- Type: ╬▒-Alkylation
---
## e)
**Given:**
- Reactant: Alcohol + Carboxylic acid
- Reagent: Acid
- Product: Ester
**Type:** Fischer Esterification
### **Step-by-step:**
1. Alcohol reacts with carboxylic acid under acidic conditions to form ester.
**Final answer:**
- Product: Ester (combine alcohol and acid)
- Type: Fischer Esterification
---
## f)
**Given:**
- Reactant: Malonic ester
- Reagents:
1. EtOŌü╗, RX
2. HŌéāOŌü║, heat
**Type:** Malonic Ester Synthesis
### **Step-by-step:**
1. Deprotonation, alkylation, hydrolysis, and decarboxylation yield substituted acetic acid.
**Final answer:**
- Product: Substituted acetic acid
- Type: Malonic Ester Synthesis
---
## g)
**Given:**
- Reactant: Ester with an ╬▒-proton
- Reagents:
1. EtOŌü╗, Ester
2. HŌéāOŌü║
**Type:** Claisen Condensation
### **Step-by-step:**
1. Enolate attacks another ester, forming ╬▓-ketoester.
**Final answer:**
- Product: ╬▓-Ketoester
- Type: Claisen Condensation
---
## h)
**Given:**
- Reactant:
- Reagents:
1. MeOŌü╗, OMe
2. HŌéāOŌü║, heat
**Type:** Dieckmann Condensation (intramolecular Claisen)
### **Step-by-step:**
1. Intramolecular condensation forms cyclic ╬▓-ketoester.
**Final answer:**
- Product: Cyclic ╬▓-ketoester
- Type: Dieckmann Condensation
---
## i)
**Given:**
- Reactant:
- Reagents:
1. HŌééO, THF
**Type:** Aldol Addition
### **Step-by-step:**
1. Enolate addition to aldehyde/ketone forms ╬▓-hydroxy carbonyl.
**Final answer:**
- Product: ╬▓-Hydroxy carbonyl
- Type: Aldol Addition
---
## j)
**Given:**
- Reactant:
- Reagents:
1. PhCHO, NaOH, EtOH
**Type:** Aldol Condensation
### **Step-by-step:**
1. Enolate addition, followed by dehydration to ╬▒,╬▓-unsaturated carbonyl.
**Final answer:**
- Product: ╬▒,╬▓-Unsaturated carbonyl (enone or enal)
- Type: Aldol Condensation
---
### **Summary Table**
| Letter | Product (structure) | Type |
|--------|---------------------------------|------------------------------|
| a | ╬▒-Bromoacetoacetate | ╬▒-Bromination |
| b | Acid chloride | Formation of acid chloride |
| c | Amide | Formation of an amide |
| d | ╬▒-Benzyl cyclohexanone | ╬▒-Alkylation |
| e | Ester | Fischer Esterification |
| f | Substituted acetic acid | Malonic Ester Synthesis |
| g | ╬▓-Ketoester | Claisen Condensation |
| h | Cyclic ╬▓-ketoester | Dieckmann Condensation |
| i | ╬▓-Hydroxy carbonyl | Aldol Addition |
| j | ╬▒,╬▓-Unsaturated carbonyl | Aldol Condensation |
If you need structures drawn out, let me know!
¤ōó Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions