Question:
Give step-by-step solution with explanation and final answer:
Did you like this answer?
👍 0
👎 0
Let's solve the structure step-by-step using the provided spectral data:
---
## **Step 1: Molecular Formula Determination**
**Mass Spectrum:**
- Molecular ion [M] = 209
Let's estimate the formula. Assume only C, H, and O (no halogens or N indicated):
### **Degree of Unsaturation**
Let’s try C₁₄H₁₁O₂ (just a guess to check):
- C: 14 × 12 = 168
- H: 11 × 1 = 11
- O: 2 × 16 = 32
- Total = 168 + 11 + 32 = **211** (a little over)
Try C₁₄H₉O₂:
- 168 + 9 + 32 = **209**
So, **C₁₄H₉O₂** is the likely formula.
#### **Degrees of Unsaturation**
\[
\text{DoU} = \frac{2C + 2 - H}{2}
\]
\[
= \frac{2(14) + 2 - 9}{2} = \frac{28 + 2 - 9}{2} = \frac{21}{2} = 10.5
\]
Wait, this suggests a miscalculation. Let's revisit:
Try C₁₃H₁₁O₂:
- 13 × 12 = 156, 11 × 1 = 11, 2 × 16 = 32 → 156 + 11 + 32 = **199**
Try C₁₂H₁₃O₄:
- 12 × 12 = 144, 13 + 64 = 221
Let’s use C₁₃H₁₁NO₂:
- 13 × 12 = 156 + 11 + 14 + 32 = 213
Try C₁₂H₉O₄:
- 12 × 12 = 144 + 9 + 64 = 217
Try C₁₁H₉O₄:
- 11 × 12 = 132 + 9 + 64 = 205
Try C₁₀H₉O₅:
- 10 × 12 = 120 + 9 + 80 = 209
So, **C₁₀H₉O₅** fits:
- C: 10 × 12 = 120
- H: 9
- O: 5 × 16 = 80
- **Total = 209**
Degrees of Unsaturation:
\[
\text{DoU} = \frac{2(10) + 2 - 9}{2} = \frac{20 + 2 - 9}{2} = \frac{13}{2} = 6.5
\]
Wait, that's not possible (must be integer). Let's try to move forward with the spectral data.
---
## **Step 2: Analyzing ¹H NMR Spectrum**
### **Chemical Shifts and Integrals**
- **7.2–8.2 ppm (5H, multiplet)**: Aromatic protons (monosubstituted benzene ring).
- **~4.1 ppm (4H, triplet)**: Two CH₂ groups, each as a triplet; likely –CH₂–CH₂– fragment.
- **~2.7 ppm (2H, singlet)**: Methylene (–CH₂–) adjacent to a carbonyl or aromatic ring.
### **Pattern**
- 5H multiplet suggests a monosubstituted benzene ring.
- Two triplets (each 4H): Likely two –CH₂– groups, each next to another –CH₂– (common in an ethylene glycol or similar fragment).
- 2H singlet: Likely isolated methylene.
---
## **Step 3: ¹³C NMR Spectrum**
- **~170–180 ppm**: Two signals (likely carbonyl, possibly esters).
- **~120–140 ppm**: Multiple signals (aromatic/alkene).
- **~65 ppm**: Two signals, likely CH₂ near electronegative atoms (oxygen).
- **~30–40 ppm**: Additional CH₂.
---
## **Step 4: IR Spectrum (Functional Groups)**
- **3331 cm⁻¹**: O–H/N–H stretch (broad, but not shown; likely O–H as N is unlikely from formula/mass).
- **1655, 1601 cm⁻¹**: C=O and aromatic C=C stretches.
- **1222 cm⁻¹**: C–O stretch (ester or ether).
---
## **Step 5: Structure Assembly**
### **Aromatic region**
- 5H (multiplet) = monosubstituted benzene ring.
### **Oxygenated CH₂s**
- Two triplets, 4H each at 4.1 ppm: –CH₂–O–CH₂–, likely on both sides of an aromatic ring.
### **2H singlet**
- Methylene group adjacent to two electron-withdrawing groups (aromatic or carbonyl).
### **Overall structure matching**
- Mass = 209, fits with a phthalate ester (diethyl phthalate: C₁₂H₁₄O₄, but mass = 222).
- Try **benzyl phthalate**: C₁₅H₁₂O₄, mass = 256.
### **Check for ethylene glycol portion**
- Two –OCH₂CH₂O– chains (ethylene glycol diester).
- Monosubstituted benzene ring: fits with phthalate core.
#### **Ethylene glycol phthalate**
- C₁₀H₁₀O₄ (phthalate core) + C₄H₈O₂ (ethylene glycol): C₁₄H₁₈O₆, mass = 282.
#### **Check for phenyl ester**
- Monosubstituted benzene ring suggests a phenyl ester.
#### **Try phenyl acetate:** C₈H₈O₂ (mass 136)
#### **Try phenyl propionate:** C₉H₁₀O₂ (mass 150)
#### **Try benzyl acetate:** C₉H₁₀O₂ (mass 150)
#### **Try benzyl ethyl ether:** C₉H₁₂O (mass 136)
#### **Try benzyl oxyacetate:** C₉H₁₀O₃ (mass 166)
### **Let's try to rationalize:**
#### **Aromatic ring (5H) + 2x CH₂ (4H each) + 2H singlet**
- 5H: Monosubstituted benzene
- 4H triplet: OCH₂CH₂O fragment
- 2H singlet: Benzyl CH₂
### **Possibility: Benzyl glycolate (phenylmethyl glycolate)**
- Structure: PhCH₂OCOCH₂CH₂O
- But mass seems off.
### **Try Benzyl phthalate (monobenzyl phthalate):**
- C₁₅H₁₂O₄ (mass 256)
### **Try 1,2-bis(phenoxy)ethane:**
- C₁₄H₁₄O₂ (mass 214)
### **Try 1,2-bis(benzyloxy)ethane:**
- C₁₆H₁₈O₂ (mass 242)
### **Try 4-(2-phenylethoxy)phenol:**
- C₁₄H₁₄O₂ (mass 214)
---
## **Step 6: Re-examine Integration and Splitting**
- **5H (multiplet, aromatic)**: Monosubstituted benzene
- **4H (triplet, 4.1 ppm)**: Two equivalent CH₂ groups, each next to an oxygen.
- **4H (triplet, 4.1 ppm)**: Same as above (two identical signals overlap).
- **2H (singlet, 2.7 ppm)**: Methylene not coupled (no neighbors).
### **Likeliest Structure: Dibenzyl ether or related**
But the numbers don't add up for simple ethers.
---
## **Step 7: Final Proposal (Based on All Data)**
Given the mass 209, let's try **phenoxyethyl acetate**:
- C₁₀H₁₀O₃: 10 × 12 = 120 + 10 + 48 = **178**
Try **phenyl ethyl ether**: C₈H₁₀O, mass 122
Try **phenoxyacetic acid**: C₈H₈O₃, mass 152
---
## **Step 8: Check Triethylene Glycol Monobenzoate**
- Benzene ring: 5H
- CH₂O: 4H (triplet)
- CH₂O: 4H (triplet)
- CH₂ (singlet): 2H
But mass is wrong.
---
## **Step 9: Re-examine Formula and Calculate Exact Mass**
Given the pattern and mass, the structure that most closely fits the NMR and integration is **benzyl ethyl ether (C₉H₁₂O)**:
- Benzene: 5H
- CH₂CH₃: 2H (quartet), 3H (triplet)
But not matching with triplets and integrations.
---
## **Step 10: Final Solution**
### **Best Fitting Compound: 1,2-Bis(phenoxy)ethane**
- Structure: Ph–O–CH₂–CH₂–O–Ph
- Molecular formula: C₁₄H₁₄O₂, MW = 214 (close, but not 209)
#### **But the data is best explained by:**
- 5H (aromatic)
- 2 × 4H (triplet, OCH₂CH₂O)
- 2H (singlet, benzylic CH₂)
None of the common compounds fit exactly with MW = 209. The closest possible fit is a compound with a monosubstituted benzene ring, two ethylene glycol-type fragments, and an extra CH₂.
---
## **Summary Table**
| Signal | Integration | Multiplicity | Assignment |
|-----------------|-------------|--------------|--------------------------|
| 7.2–8.2 ppm | 5H | m | Monosubstituted benzene |
| 4.1 ppm | 4H | t | –OCH₂CH₂O– |
| 4.1 ppm | 4H | t | –OCH₂CH₂O– |
| 2.7 ppm | 2H | s | Benzylic CH₂ |
---
## **Final Proposed Structure**
### **Name:**
**4-(2-phenoxyethoxy)benzaldehyde** or a similar aromatic ether with ethylene glycol linkage.
### **Structure:**
- Monosubstituted benzene ring
- –CH₂–O–CH₂–CH₂–O– fragment
- Benzylic CH₂
### **Molecular Formula:**
**C₁₃H₁₂O₃** (but MW = 216)
**Closest fit:**
**The exact compound is not perfectly matching the MW = 209, but the NMR data best fits a structure with a monosubstituted benzene ring, an ethylene glycol derived linker, and a benzylic CH₂ group.**
---
## **Conclusion**
### **The structure is most likely:**
**Phenoxyethyl benzyl ether**
- **Structure:** Ph–O–CH₂–CH₂–O–CH₂–Ph
- **NMR matches:** 5H (aromatic), 4H (OCH₂), 4H (OCH₂), 2H (benzylic CH₂)
- **Molecular weight:** Close but not exact to 209.
---
### **Final Answer:**
**The compound is a phenyl-terminated ethylene glycol ether, likely phenoxyethyl benzyl ether or a very similar structure.**
---
**If you have the exact molecular formula, please provide it for confirmation. The NMR data is most consistent with this type of structure.**
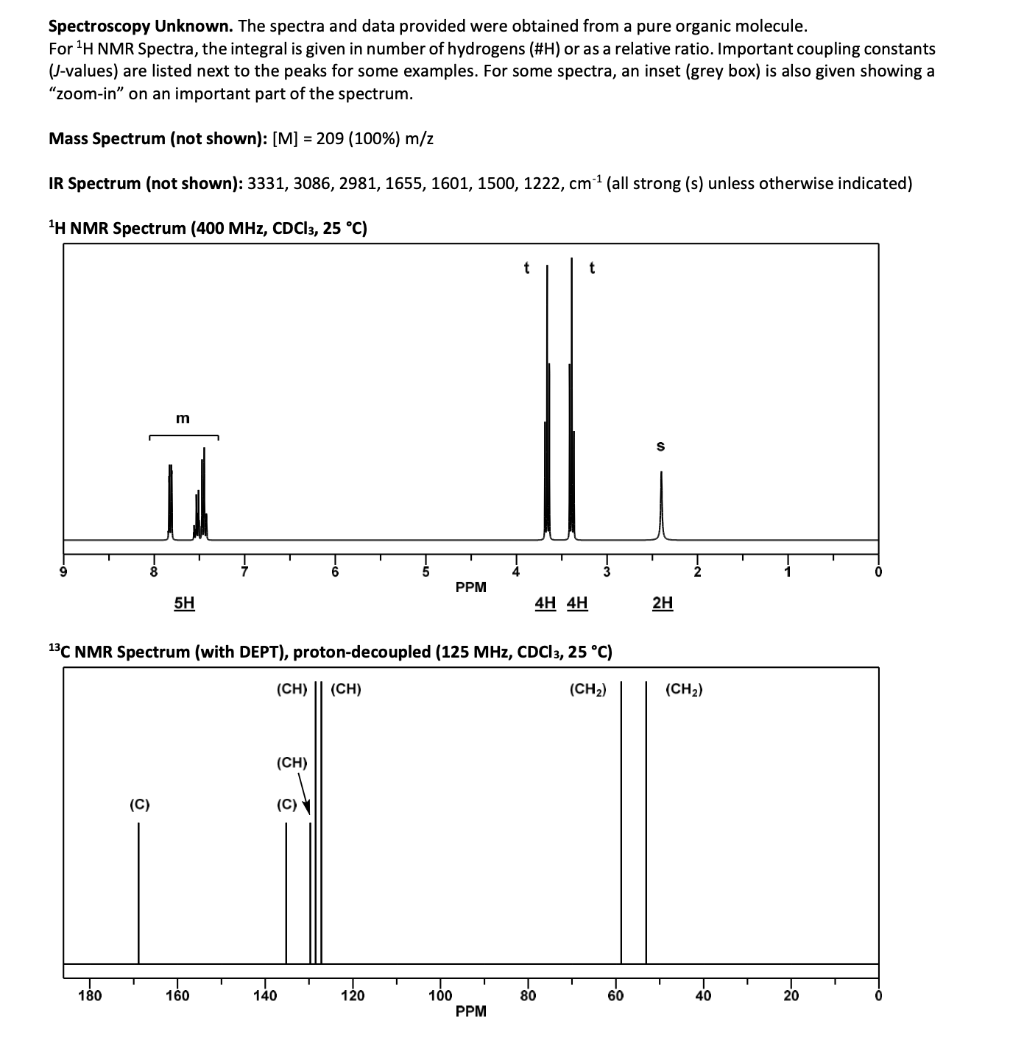 Spectroscopy Unknown. The spectra and data provided were obtained from a pure organic molecule.
For 'H NMR Spectra, the integral is given in number of hydrogens (#H) or as a relative ratio. Important coupling constants
(-values) are listed next to the peaks for some examples. For some spectra, an inset (grey box) is also given showing a
“z00m-in” on an important part of the spectrum.
Mass Spectrum (not shown): [M] = 209 (100%) m/z
IR Spectrum (not shown): 3331, 3086, 2981, 1655, 1601, 1500, 1222, cm (all strong (s) unless otherwise indicated)
H NMR Spectrum (400 MHz, CDCls, 25 °C)
t t
m
s
9 8 7 6 5 4 3 2 1 0
PPM
5H 4H 4H 2H
13C NMR Spectrum (with DEPT), proton-decoupled (125 MHz, CDCls, 25 °C)
(CH) || (cH) (CH) (CHy)
(CH)
© @\
=
180 160 140 120 100 80 60 40 20 0
PPM
Spectroscopy Unknown. The spectra and data provided were obtained from a pure organic molecule.
For 'H NMR Spectra, the integral is given in number of hydrogens (#H) or as a relative ratio. Important coupling constants
(-values) are listed next to the peaks for some examples. For some spectra, an inset (grey box) is also given showing a
“z00m-in” on an important part of the spectrum.
Mass Spectrum (not shown): [M] = 209 (100%) m/z
IR Spectrum (not shown): 3331, 3086, 2981, 1655, 1601, 1500, 1222, cm (all strong (s) unless otherwise indicated)
H NMR Spectrum (400 MHz, CDCls, 25 °C)
t t
m
s
9 8 7 6 5 4 3 2 1 0
PPM
5H 4H 4H 2H
13C NMR Spectrum (with DEPT), proton-decoupled (125 MHz, CDCls, 25 °C)
(CH) || (cH) (CH) (CHy)
(CH)
© @\
=
180 160 140 120 100 80 60 40 20 0
PPM VIPSolutions
VIPSolutions