Give step-by-step solution with explanation and final answer:382% EEE giz ÂŁ21 9 Ere i: ÂŁ4
Question:
Give step-by-step solution with explanation and final answer: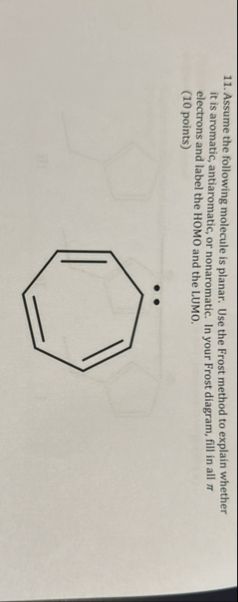 382%
EEE
giz
ÂŁ21
9 Ere
i:
ÂŁ4
382%
EEE
giz
ÂŁ21
9 Ere
i:
ÂŁ4
This is AI generated Response.
Asked by: Rahul
Created at: 2026-02-23 04:14:09
Asked by: Rahul
Created at: 2026-02-23 04:14:09
Final Answer:
Rendered
Raw Text
Did you like this answer?
Let's solve the problem step by step:
---
## **Step 1: Identify the Mole**
The molecule shown is cycloptatrienyl anion (often called the "tropylium anion"). It is a seven-membered ring with three double bonds and a pair of lone electrons (from the negative charge).
---
## **Step 2: Count the π Electrons**
- Each double bond contributes **2 π electrons**.
- There are **3 double bonds**: \(3 \times 2 = 6\) π electrons.
- The lone pair on the carbon (from the negative charge) adds **2 more π electrons**.
**Total π electrons:**
\[
6 + 2 = 8
\]
---
## **Step 3: Apply the Frost Circle (Polygon) Method**
The Frost circle helps us determine the molecular orbitals for planar, cyclic, conjugated systems:
- Draw a regular heptagon (7 sides) with one vertex pointing down.
- Inscribe this polygon in a circle.
- Each vertex represents a π molecular orbital.
- The horizontal axis passing through the center is the reference for energy.
#### **Energy Levels:**
For 7-membered ring:
- 1 orbital at the bottom (non-degenerate, lowest energy)
- 2 pairs of degenerate orbitals (next higher energy)
- 2 orbitals at the top (highest energy, degenerate)
---
## **Step 4: Fill the Molecular Orbitals**
Fill the π electrons (8 total) from the lowest energy upwards, **two electrons per orbital**:
1. **Lowest MO:** 2 electrons (filled)
2. **Next degenerate pair:** 4 electrons (2 per orbital, filled)
3. **Next degenerate pair:** 2 electrons (each orbital gets 1 electron, both half-filled)
So, the 8th electron goes into the next orbital, resulting in **two half-filled degenerate orbitals**.
---
## **Step 5: Aromaticity Analysis (HĂĽckel's Rule)**
- **Hückel's Rule:** A molecule is aromatic if it is planar, fully conjugated, and contains \(4n + 2\) π electrons (where \(n\) is a non-negative integer).
- Here, π electrons = 8.
- \(4n + 2 = 8 \implies n = 1.5\), which is **not** an integer.
**Therefore:**
- The molecule is **not aromatic**.
- It is **antiaromatic** if it is planar and fully conjugated with \(4n\) π electrons (\(n=2\) here).
- **8 π electrons fit \(4n\) (n=2): Antiaromatic.**
---
## **Step 6: Draw Frost Diagram and Label HOMO/LUMO**
```
o (highest energy, half-filled)
o o (second highest, half-filled)
o o (middle, filled)
o o (lowest, filled)
o (lowest, filled)
```
- **HOMO (Highest Occupied Molecular Orbital):** The two half-filled degenerate orbitals.
- **LUMO (Lowest Unoccupied Molecular Orbital):** The next available level above the half-filled orbitals.
---
## **Step 7: Final Answer**
### **Summary Table**
| Property | Value |
|-----------------|---------------------------------------|
| π electrons | 8 |
| Frost Diagram | Fill from bottom, 2 electrons per MO |
| HOMO | Two degenerate half-filled orbitals |
| LUMO | Next higher unfilled orbital |
| Aromatic? | **No** (not \(4n+2\) π electrons) |
| Antiaromatic? | **Yes** (\(4n\) π electrons, planar) |
| Nonaromatic? | No (because it's planar and conjugated)|
---
## **Conclusion**
> **The molecule has 8 π electrons and is antiaromatic according to Hückel’s rule. The Frost circle shows two half-filled degenerate HOMOs. The molecule is not aromatic.**
---
### **Diagram (Frost Circle)**
Below is a schematic Frost circle for a 7-membered ring, filled with 8 electrons:
```
o
o o
o o
o o
o
```
Fill electrons from the bottom up (2 per orbital), label the HOMO as the two half-filled orbitals, and the LUMO as the next empty orbital above.
---
If you need an explicit energy diagram drawn or further clarification, let me know!
📢 Instructions:
- Ask anything relatd to above question only.
- For new Question goto :
 VIPSolutions
VIPSolutions